 
How to measure density of liquid petroleum productsĮxample 2: Using Density to Identify a MaterialĮxample 3: Density to Calculate Volume Mass Water - Density, Specific Weight and Thermal Expantion Coefficient - variation with temperature at 1, 68 and 680 atm, SI and Imperial unitsĪir - Density, Specific Weight and Thermal Expantion Coefficient - variation with temperature and pressure, SI and Imperial units Se also: Densities for some common materials Density is a physical property - constant at a given temperature and pressure - and may be helpful for identification of substances.īelow on this page: Specific gravity (relative density), Specific gravity for gases, Specific weight, Calculation examples On atomic level - particles are packed tighter inside a substance with higher density. See also Unit converter - mass and Unit converter - density Slugs can be multiplied with 32.2 for a rough value in pound-mass (lb m). 
Note that there is a difference between pound-force (lb f) and pound-mass (lb m). The Imperial (U.S.) units for density are slugs/ft 3 but pound-mass per cubic foot - lb m/ft 3 - is often used. What is weight and what is mass? - the difference between weight and mass.Mass is a property and the SI unit for density is. The water density calculator might give you more insights into this problem.Density is defined as mass per unit volume. Every dissolved particle inside a body of water affects its density. It changes slightly whether it is tap, fresh, or salt water. There are other aspects that affect water density. If water froze from the bottom up, then life never would have gotten the chance. It is this same principle that scientists think helped life get started on Earth. This phenomenon, coupled with a low thermal conductivity of ice, helps the bottom of the lake stay unfrozen, so that fish can survive. The colder water stays at the surface and turns to ice. Since the water at 4 ☌ is the heaviest, it falls to the bottom of the lake. How's this important? It makes it much harder for lakes to freeze completely in the winter. However, at approximately 4 ☌ degrees, water reaches its maximum density. If you cool water from room temperature, it becomes increasingly dense. While the general rule is that as temperature goes up, the density lowers, water behaves differently between 0 ☌ and 4 ☌. However, we have a slight, but a super important anomaly when it comes to water. However, as with almost all materials, its density changes with temperature. We have prepared the grams to cups calculator specifically for that purpose.Īllow us to throw in a bit of a curve ball here by reminding you that if you want to calculate the density of pixels on your screen, this is not the calculator you are looking for try PPI calculator instead.įor most purposes, it's enough to know that the density of water is 1,000 kg/m 3. If you know the density of the product as well as its weight in grams, you can find the volume of the ingredient in cups. Sometimes people are looking to convert grams into cups. Plug in your result there the tool will convert it into: If your unit is not there, you can use our density conversion tool. If all you need is to convert between different units, just click on the units for density and select your desired units from the list. After typing these values into the density calculator, it will give you the result in kilograms per cubic meter. Make a note of the object's weight and volume. 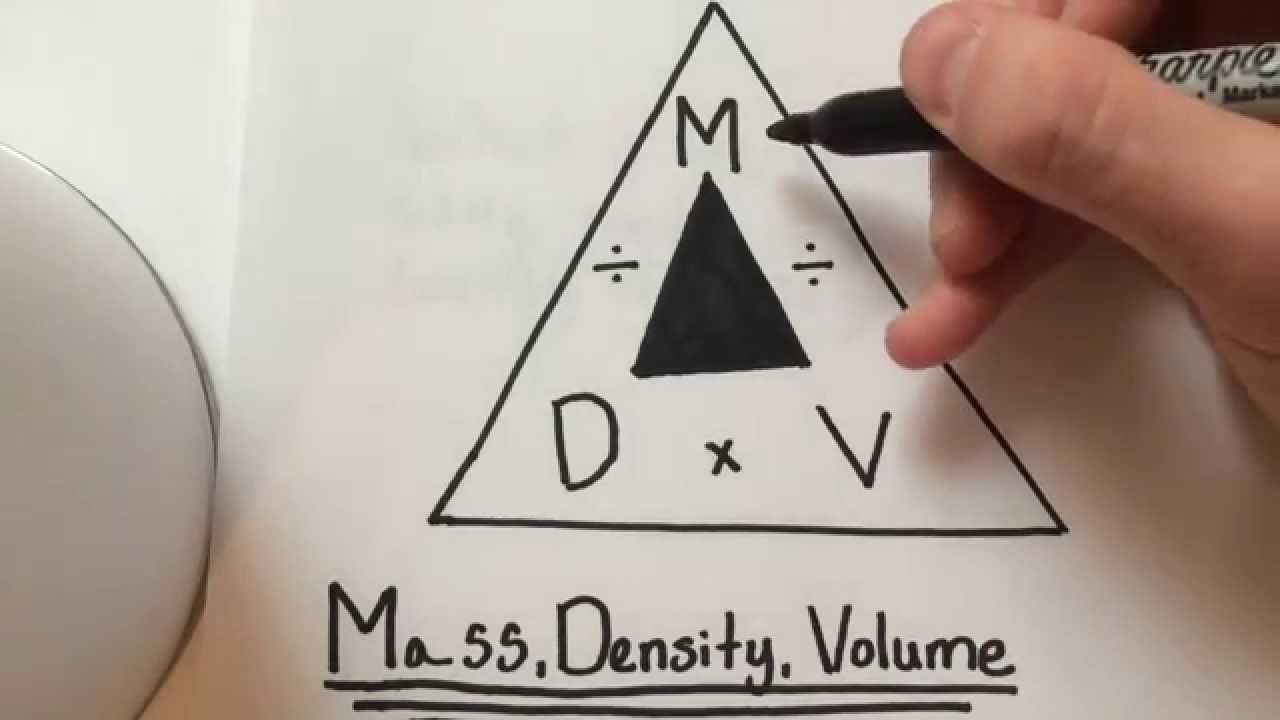
To make the calculation, you'll need to know a few other values to start with. The fastest way to find the density of an object is, of course, to use our density calculator. Or you can use our density calculator to make it a breeze! 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
